The lotion stabilized by solid particles is called Pickering lotion. Chitin is a renewable natural polysaccharide with a content second only to cellulose in nature. It is widely present in the exoskeletons of shrimp, crabs, and other crustaceans, and has good biocompatibility and biodegradability. Chitin also has the effects of lowering blood pressure, lowering blood lipids, improving gastrointestinal function, and regulating the body's immune system, and can play an important role in the field of health food. In addition, chitin molecular chains contain a large amount of - OH and - NHCOCH3, which have good amphiphilicity and are easy to modify. Chitin nanofibers (ChNFs) are one-dimensional nanomaterials prepared from chitin, which not only possess the excellent properties of chitin, but also have high specific surface area, crystallinity, and modulus. They are a highly promising food grade Pickering emulsifier. ChNFs and chitin nanocrystals can effectively stabilize O/W Pickering lotion.
Dong Tongjun, Wei Kongju and Ding Beibei * from the School of Food Science and Engineering of Wuhan University of Light Industry purified, partially deacetylated and carboxymethyl chitin to obtain ChNFs with both amino and carboxyl groups, characterized its structure and used it as an emulsifier to stabilize Pickering lotion, evaluated the stability of lotion in combination with rheological properties, laser confocal microscope, etc., and observed its storage stability and adaptability to pH value. In order to protect and deliver bioactive substances, especially pH sensitive substances, in the fields of food and biotechnology.

Morphological characterization of ChNFs and RE ChNFs
Figure 1A shows the atomic force microscopy (AFM) images of ChNFs and RE ChNFs aqueous dispersions, respectively, which clearly demonstrate the fiber morphology of individual nanochitin nanoparticles. Moreover, freshly prepared and redispersed nanochitin is well dispersed in aqueous media without any aggregation phenomenon. Statistical analysis was conducted on the length and width of two types of nanofibers using image processing software, as shown in Figures 1B and C. The average length and diameter of ChNFs before and after redispersion were 271.2 nm and 295.4 nm, 39.4 nm and 44.5 nm, respectively, with aspect ratios of approximately 6.9 and 6.7, respectively. The lower aspect ratio also conforms to the "short and fat" morphology of α - chitosan. In addition, a few shorter substances were observed in the AFM image. Unlike some published research results, the size changes of nanofibers in the prepared ChNFs suspension and redispersion suspension are not significant. This may be due to the short ultrasonic treatment time during the preparation process, which has little or even negligible effect on the size of chitosan. Figure 2A shows the appearance of ChNFs aqueous dispersion before and after redispersion at the same concentration. The results showed that when the concentration of chitin in the aqueous dispersion was the same, the transparency difference between freshly prepared ChNFs and RE ChNFs aqueous dispersion was not significant, that is, the turbidity was similar. The ChNFs obtained by drying and removing the dispersion medium can be redispersed in aqueous medium by simple ultrasonic treatment. Compared with freshly prepared samples, the process of redispersing after removing the dispersion medium does not affect the dispersibility of ChNFs. This is mainly because chitosan greatly weakens the hydrogen bonding between molecules after treatment with concentrated alkali and carboxymethylation reagents.

The AFM and TEM results (Figure 2B) indicate that the use of chloroacetic acid to treat DE chitin can yield redispersible nanochitin, and the process of freeze-drying to remove the water dispersion medium and ultrasonic treatment during redispersion has little effect on the length and width of nanochitin, indicating that freeze-drying and brief ultrasonic treatment during redispersion do not affect the morphology of chitin.
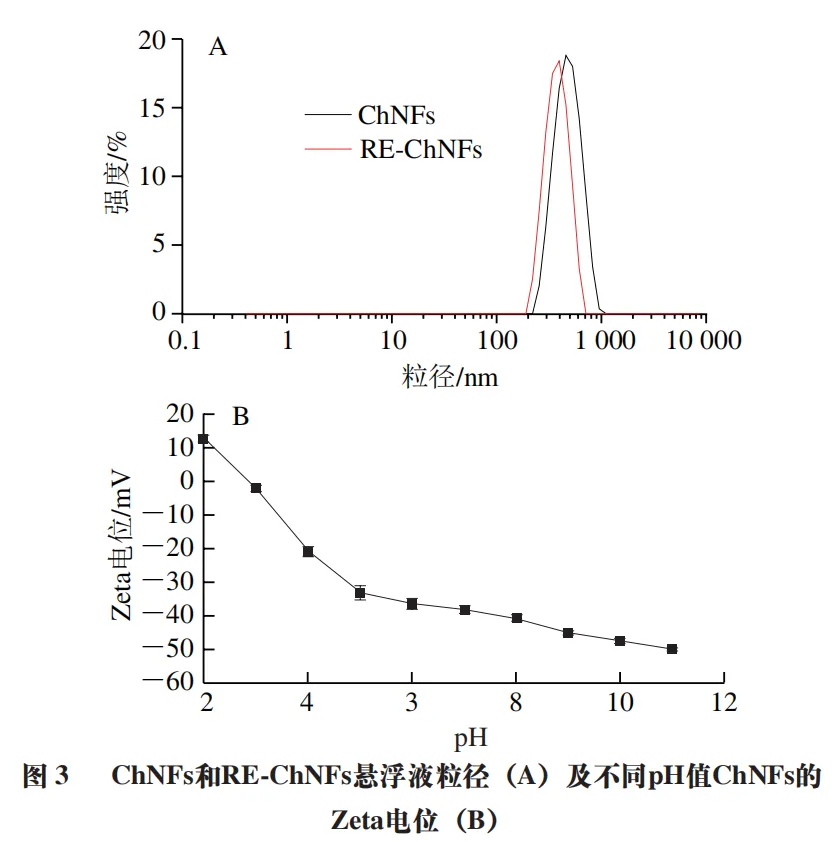
The particle size distribution of freshly prepared and redispersed ChNFs suspensions is shown in Figure 3A. Prior to measurement, the suspension was appropriately diluted with ultrapure water to avoid the influence of multiple scattering phenomena. The results showed that the particle size distribution of freshly prepared and redispersed ChNFs suspensions was basically the same, indicating that the redispersion process did not have a significant impact on the morphology of chitin. This is consistent with the conclusions obtained from AFM and TEM. According to Figure 3B, the Zeta potential value of ChNFs suspension at pH 7 is - (38.1 ± 1.2) mV, with an absolute value greater than 30, and the polydispersity index (PDI) is 0.380, indicating uniform particle size distribution. In addition, according to the intensity of particle size distribution, it can be seen that the size of the ChNFs and RE ChNFs suspensions produced is relatively uniform, with a small particle size range, which is consistent with the smaller PDI values measured.
Structural characterization of 2 ChNFs and RE ChNFs
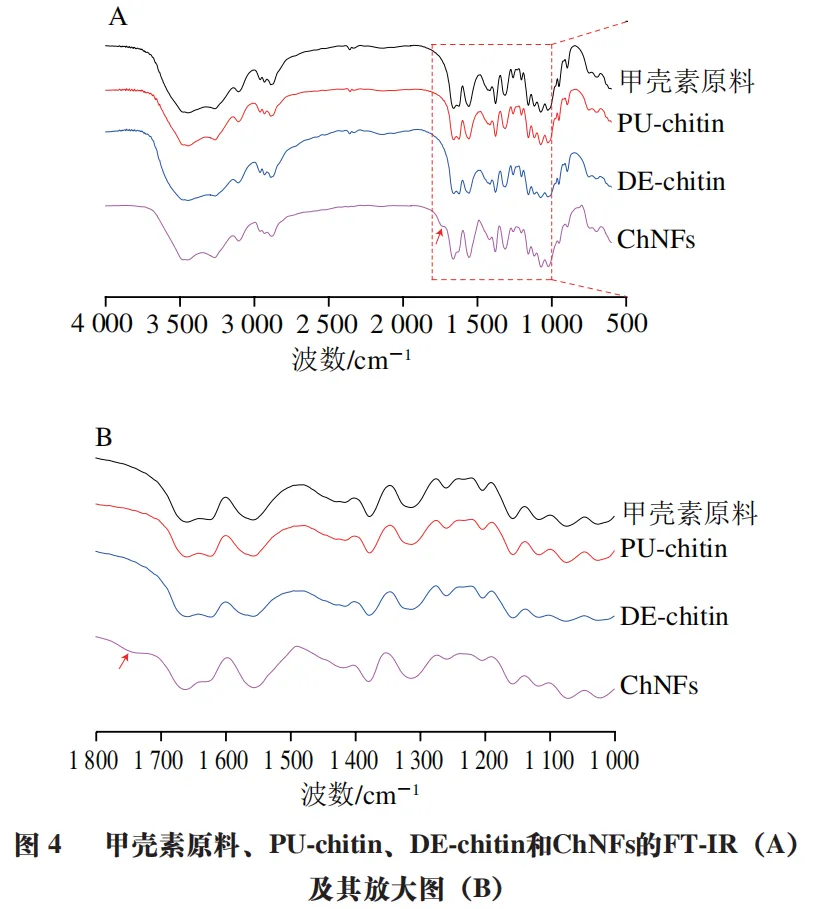
Determine the chemical and crystal structure of the prepared ChNFs through FT-IR and XRD results. Figure 4A shows the infrared images of chitin raw materials, PU chitin, DE chitin, and ChNFs. Figure 4B is an enlarged image in the range of 1800 to 1000 cm-1. It can be seen that a small peak appears near 1730 cm-1, which is a characteristic peak of carboxyl groups, indicating the successful introduction of carboxyl groups into the chitin molecular chain. Similar to the results of chitin raw materials and PU chitin, the amide I bond of ChNFs exhibits dual absorption peaks at 1660 cm-1 and 1621 cm-1, indicating that the crystal structure of chitin used in the experiment is in the α - form and has not changed throughout the entire processing.
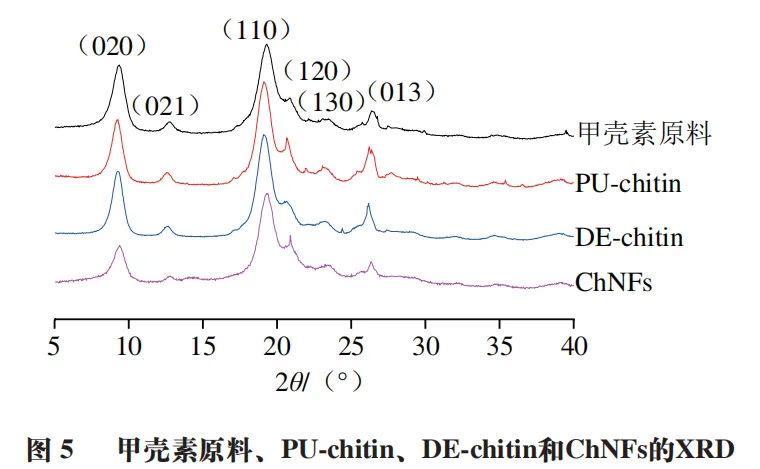
The XRD results in Figure 5 show that all four types of chitin samples exhibit diffraction peaks at 9.4 °, 12.8 °, 19.3 °, 20.8 °, 23.4 °, and 26.4 °, corresponding to the (020), (021), (110), (120), (130), and (013) crystal planes in the α - chitin crystal, which is consistent with the FT-IR spectrum results. Meanwhile, FT-IR spectra and XRD results indicate that during the preparation of ChNFs, partial deacetylation and carboxymethylation only occur on the surface of ChNFs. Compared with untreated chitin, during the purification, partial deacetylation, and carboxymethylation of chitin, the peak values of the sample crystallization peaks gradually decreased with the increase of the treatment process, indicating that the intermolecular hydrogen bonds of chitin were further disrupted during the treatment process.
3 Rheological properties of lotion
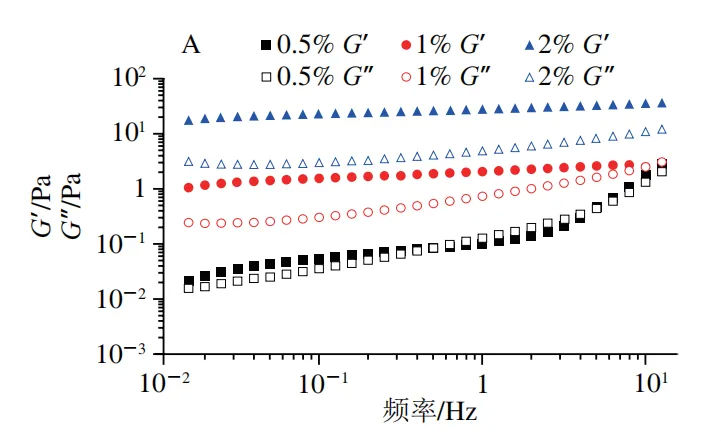
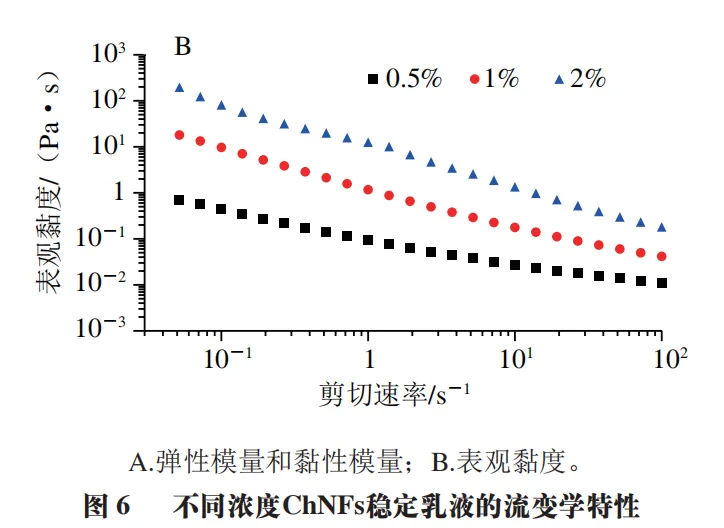
Figure 6A shows the change trend of the elastic modulus (G ′) and viscous modulus (G ″) of Pickering lotion with frequency. It can be seen that even if the content of ChNFs in the lotion is very low, G ′ of the three samples is higher than G ″ in the low-frequency zone, indicating that these lotion all show obvious gel characteristics. When the mass fraction of ChNFs is greater than 1.0%, G ′ and G ″ values of the stabilized lotion samples do not depend on the change of frequency in the low-frequency zone. In addition, when the mass fraction of ChNFs is 1.0% and 2.0%, the change trend of G ′ and G ″ of the stable Pickering lotion is slightly different from that of 0.5%. When the mass fraction of ChNFs used to stabilize the lotion is low (0.5%), G ′ and G ″ are low at lower frequencies and intersect at higher frequencies. This phenomenon is similar to the research results of Xu Baomei, who attributed this phenomenon to the fact that there are few or no network structures in the lotion. With the increase of the mass fraction of ChNFs in the stable lotion, the G ′ and G ″ of the lotion gradually increase. When the mass fraction of ChNFs in the lotion is 2.0%, the G ′ and G ″ values of the lotion are about 22 Pa and 3 Pa, respectively. At this time, the G ′ value exceeds the G ″ value by more than 7 times, and remains relatively unchanged in the frequency range of 0.1 to 10 Hz, indicating that the substances in the lotion form a gel like network structure at this time. Figure 6B shows the variation trend of the viscosity of Pickering lotion with the shear rate. It can be seen that when the shear rate is higher than 0.1 s-1, the three lotion samples all show typical shear thinning behavior, that is, at low shear rates, the shear viscosity of lotion is very high; With the increase of shear rate, the shear viscosity of lotion gradually decreases, which indicates that the lotion network can be destroyed by a higher shear rate, and this phenomenon also proves that the lotion prepared belongs to oil in water lotion. In addition, the apparent viscosity of lotion increases with the increase of ChNFs mass fraction, which may be caused by the increase of viscosity of continuous phase of ChNFs suspension.
4 Microstructure of lotion
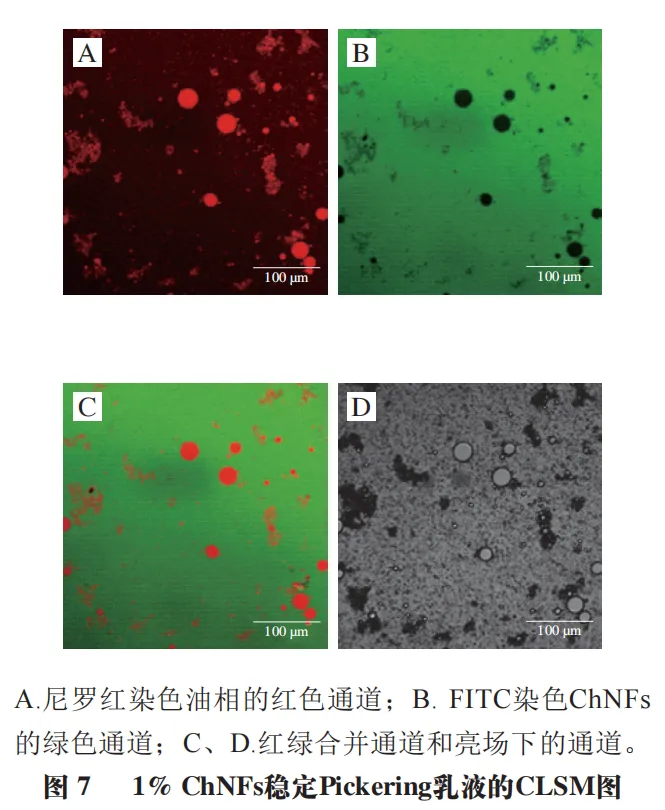
Figure 7 shows the CLSM image of the Pickering lotion that is stable when the mass fraction of ChNFs is 1.0%. The oil phase is dyed with Nile red, and ChNFs is dyed with FITC. It can be seen that the green ChNFs in the lotion almost occupy the whole observation background area, showing a continuous phase distribution (Figure 7B), while the red paraffin shows a relatively independent droplet distribution (Figure 7A). As shown in Figure 7C, the water continuous phase of ChNFs is surrounded by independently distributed oil droplets. This phenomenon confirms that the prepared lotion is O/W lotion, which is also consistent with the rheological test results of lotion.
5 Storage stability of lotion

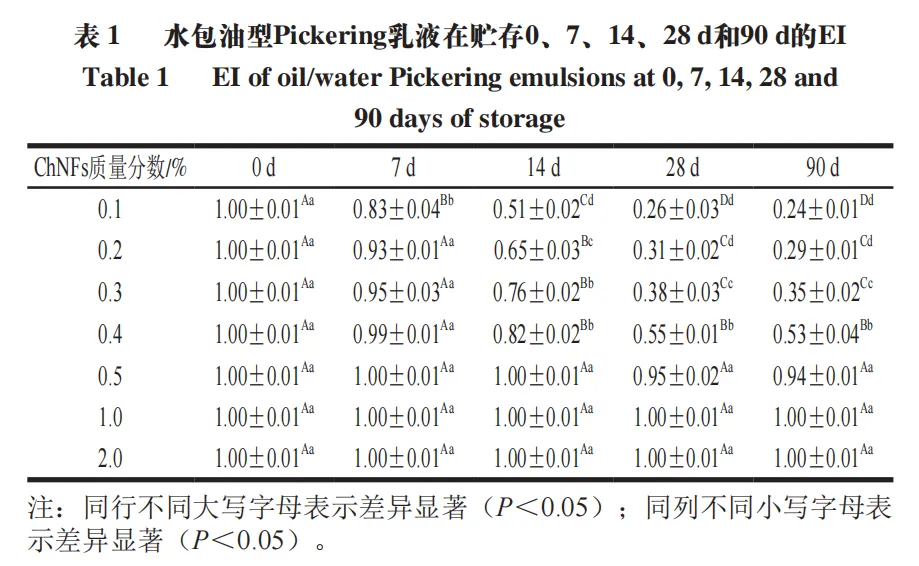
As shown in Figure 8A, with the increase of the mass fraction of ChNFs in the dispersion, the light scattering of ChNFs dispersion increases, that is, the turbidity increases. Figures 8B to F show the appearance pictures of Pickering lotion stabilized with different mass fractions of ChNFs stored at room temperature for 0, 7, 14, 28 and 90 days. Table 1 shows the EI of different lotion samples during storage. These results show that 0.1% ~ 2.0% ChNFs can effectively stabilize Pickering lotion at 0 d (fresh preparation), and there is no significant difference in EI value. In addition, when the mass fraction of ChNFs used to stabilize the lotion is low (0.1%, 0.2%), the sample will show obvious delamination during the storage period of 0~7 days, and a small amount of delamination can be observed at the bottom of the lotion after 3 days. With the extension of the storage time, the delamination gradually becomes obvious. On the contrary, when the mass fraction of ChNFs used to stabilize lotion is high (0.5% ~ 2.0%), the prepared lotion will not show obvious stratification during the entire 90 day storage period. Except for the 0.5% ChNFs stabilized lotion, the EI value of the other two lotion samples can be maintained at about 1 in the late storage period, indicating that the emulsification effect is good and the lotion has excellent storage stability. At the same time, it also proves that the prepared ChNFs has the potential as an emulsifier.
6 Effect of pH value on stability of lotion

By partially deacetylating ChNFs, a certain amount of amino groups are added to their surface, and then carboxymethylation is applied to give them carboxyl groups, giving ChNFs a protein like double charge property. The paraffin water lotion prepared with 2.0% ChNFs suspension at different pH values was placed for 24 hours to study the effect of pH value on the stability of lotion, in which the volume ratio of oil to water was 1 ∶ 9. It can be seen from Figure 9 that when the pH value of lotion is within the range of 3~11, ChNFs can stabilize lotion within 24 h. If the pH value of lotion is further increased or decreased, demulsification will occur. The reason for the occurrence of emulsion breaking phenomenon at higher or lower pH values may be that the nanoparticle charge density is high at this time, leading to an increase in repulsion and the phenomenon of oil-water stratification. Nevertheless, ChNFs still have the ability to stabilize lotion in a wide range of pH values. Compared with traditional pH responsive emulsifiers, the ChNFs prepared in this experiment also have the advantages of wide source, redispersibility and high biocompatibility, and have a promising application prospect.
7 Conclusion
Using deacetylation combined with carboxymethylation treatment, ChNFs containing both amino and carboxyl groups were prepared, and they can still be dispersed again after drying and removing the dispersion medium, making them convenient for large-scale transportation and storage. In addition, this ChNFs can effectively stabilize the O/W Pickering lotion. The results show that the Pickering lotion has good gel properties, good storage stability and pH adaptability in a wide range, which can be used to protect and deliver pH sensitive active substances and expand the application of chitin in the field of food biology. The preparation of redispersible chitin nanofibers and their application in stable Pickering lotion comes from Food Science, Vol. 44, No. 24, 2023, pp. 1-8 Authors: Dong Tongjun, Wei Kongju, Shuangyuan, Ding Beibei, Wang Xuedong, Ding Wenping DOI: 10.7506/spkx1002-6630-20230221-193. Click below to read the original text to view the relevant information of the article.